 Because many of its properties were intermediate between those of chlorine and iodine, Balard initially thought he had isolated a compound of the two (perhaps ICl). A deep purple vapor was released, which had a biting aroma similar to that of Scheele’s “compound.” The purple substance was identified as a new element, named iodine from the Greek iodes, meaning “violet.” Bromine was discovered soon after by a young French chemist, Antoine Jérôme Balard, who isolated a deep red liquid with a strong chlorine-like odor from brine from the salt marshes near Montpellier in southern France. 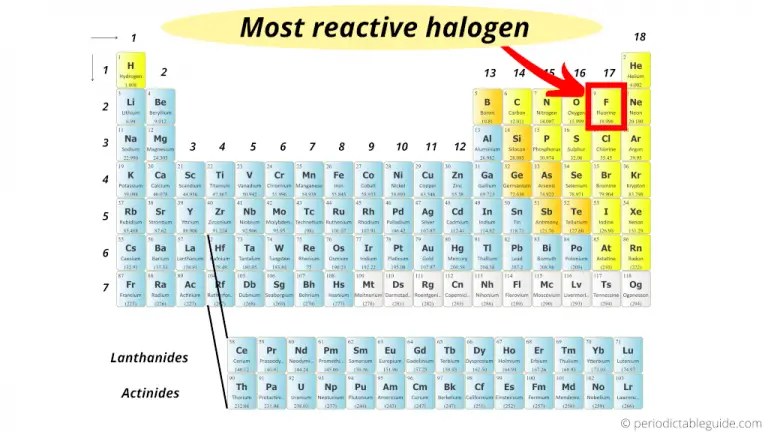
That same year, a French industrial chemist, Bernard Courtois, accidentally added too much sulfuric acid to the residue obtained from burned seaweed. In 1811, Scheele’s “compound” was identified as a new element, named from the Greek chloros, meaning “yellowish green” (the same stem as in chlorophyll, the green pigment in plants). Scheele was convinced, however, that the pale green gas he collected over water was a compound of oxygen and hydrochloric acid. In 1774, Carl Wilhelm Scheele (the codiscoverer of oxygen) produced chlorine by reacting hydrochloric acid with manganese dioxide. Despite their presence in familiar substances, none of the halogens was even recognized as an element until the 19th century.īecause the halogens are highly reactive, none is found in nature as the free element.Ĭhlorine was the first halogen to be obtained in pure form. Hydrochloric acid, which is a component of aqua regia (a mixture of HCl and HNO 3 that dissolves gold), and the mineral fluorspar (CaF 2) were well known to alchemists, who used them in their quest for gold.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
